

This article is a comprehensive and concise guide to understanding contamination of water sources, especially by pathogens and microbes.
It paints a global picture of water contamination and its consequences and delves directly into various pathogens that can be present in water and how they can harm us. Standard water management strategies to solve the issues associated with poor drinking water are described.
Special attention has been given to the concept of water quality monitoring and the different tests that have been developed to characterize water samples.
Towards the end of this article, standard methods for detection and quantification of E.coli and Total Coliforms as standard indicators of microbiological contamination of water are explained.
Finally, it describes strategies and requirements for more efficient water quality monitoring, and how new technologies that can deliver water test data onsite and in real-time can revolutionize water quality monitoring programs.

Water may make up 71% of the surface of planet earth, but only 2.5% of this water is freshwater.
Fresh water is water that is not salty and is found in lakes, rivers or streams as opposed to salt water from the oceans. With a good portion of fresh water being in glaciers, polar ice caps, the atmosphere, soil, underground in the form of groundwater or too polluted to consume, only 0.5% of the earth’s water is available to use (1). These facts emphasize that water supplies are indeed finite and that water is undeniably our most valuable resource.
In 2010, the United Nations Human Rights Committee declared the “human right to safe drinking water and sanitation” (2). For water to be deemed acceptable and safe for drinking, many factors are taken into consideration from its clarity, taste, and chemical composition to the presence of microorganisms.
Around the world more than 2 billion people use a drinking water source that is contaminated with feces causing about 829,000 deaths per year due to unsafe drinking water (3). Diarrheal disease in general claims the lives of roughly 5,000 children every day (4) worldwide, particularly in developing countries. The leading cause of diarrheal death in children is unsafe drinking water with almost 1,200 deaths per day (5).

The significance of the impact of water contaminated with disease causing microorganisms on human health can not be overstated. From the adverse effects of cholera on skin, to diarrhea, dysentery and loss of life, micro-biologically polluted water is dangerous.
Although the statistics presented here are not distributed equally around the world and developing countries form a large percentage of the affected population, water safety in developed countries should not be overlooked. In order to prevent life threatening conditions arising from unsafe drinking water, regular and frequent water testing procedures alongside boil water advisories have been implemented over the years. Boil water advisories are issued when water is known not to be safe or may be unsafe (6).
In North America, boil water advisories are commonplace, particularly in certain rural areas with poorer socio-economic communities, and governments are increasingly focused on eliminating these advisories and ensuring the right to safe water for all citizens.
There are several issues arising from having to boil drinking water with physical and mental implications. Boil water advisories, especially long term ones can hinder community trust in the quality of water. They can put financial burdens on communities if families have to rely on buying bottled water. Excessive water boiling can lead to the presence of mould in housings and force overcrowding of people, particularly in indigenous communities.
Overcrowding as well as the lack of running lead to contraction of methicillin-resistant Staphylococcus aureus bacteria (MRSA). MRSA is the main antibiotic-resistant infection that affects Indigenous people disproportionately (7). At any given time, one in five indigenous communities in Canada is under a form of drinking water advisory (8)
References
1.United States bureau of reclamation, found at https://www.usbr.gov/mp/arwec/water-facts-ww-water-sup.html.
2.United Nations Human Rights, found at https://www.ohchr.org/en/issues/escr/pages/water.aspx.
3.World Health Organization, drinking water, found at https://www.who.int/news-room/fact-sheets/detail/drinking-water
4.UNICEF press release, found at https://www.unicef.org/media/media_19974.html.
5.Our world in data, found at https://ourworldindata.org/childhood-diarrheal-diseases.
6.Government of Canada drinking water advisories, found at https://www.sac-isc.gc.ca/eng/1538160229321/1538160276874
7. The conversation, Water crisis in First Nations communities runs deeper than long-term drinking water advisories, Corinna Dally-Starna, found at https://theconversation.com/water-crisis-in-first-nations-communities-runs-deeper-than-long-term-drinking-water-advisories-148977
8. Department of Geography and Planning, University of Saskatchewan, Saskatoon,
Indigenous perspectives on water security in Saskatchewan, Canada, Water 2020, 12(3), 810, found at https://doi.org/10.3390/w12030810

Water pollution occurs whenever a substance, such as chemicals or microorganisms, is released into water via the ocean, streams, lakes, rivers, estuaries or groundwater and interferes with the beneficial use of the water or the natural function of the ecosystem (1).
It may also be considered water pollution when energy in the form of radioactivity or heat is released into bodies of water.
Various substances can pollute water bodies in the form of the following contaminants:
These contaminants can be in the form of pathogenic microorganisms, toxic chemicals, petroleum, putrescible organic waste, sediments, plant nutrients (mostly nitrogen and phosphorus) and radioactive materials (1,2).
Emerging contaminants such as pharmaceuticals and personal care products as well as microplastics also cause water pollution.
Water pollution is not distributed equally around the world. More than 2 billion do not have access to safe drinking water and 159 million people drink water directly from untreated surface water sources like streams or lakes (4).
According to the United Nations Environment Program (UNEP) while many developed countries deal with eutrophication (excessive availability of nutrients that leads to dense growth of plants), heavy metals, emerging contaminants, nitrates and land degradation due to salinity, their water quality has significantly improved over the years.
Developing countries however are facing increasing water pollution as urban populations grow, the consumption of everyday living materials increases and more untreated wastewater is released into the environment (5).


Drinking water contaminants pose a harm to public health. As stated, biological or microbial contaminants are the most dangerous pollutants and responsible for water borne diseases and deaths all around the world. There were 16.97 deaths per 100,000 people in the world from unsafe water sources reported in 2017.
Dirty water causes the death of one human being every 10 seconds (7) and
up to 18 million cases of waterborne diseases are reported in North America every year (8).
It is worth noting that even in developed countries factors such as race, ethnicity and income can play a role on availability of safe water.

Microorganisms such as
Untreated municipal wastewater is the most dangerous contributor to water ecosystems in terms of excess nutrients and microorganisms.
A startling 80% of the world’s wastewater, mostly untreated, is released into the environment (3).

Water stress means the demand for water exceeds the amount available for use or the quality is poor and restricts its use. Water stress is exacerbated by:
The UN designated its Sustainable Development Goal (SDG) 6 to achieve universal and equitable access to safe and affordable water for all by 2030. That is why management of water sources is becoming increasingly important. Reuse and recycling of wastewater (especially for irrigation), water quality monitoring (including water testing), reversing climate change and conserving water as a few of the steps we can take to battle water stress in the world. Better water management and consequently making water available to everyone will also have a positive impact on the world economy. 263 million people have to spend at least 30 min per round trip to collect water (3).
This time can be allocated to more productive activities as well as reduce the health risk on individuals. Access to improved sources of water for children who are at the highest risk of water related diseases can result in better health, potentially lead to better school attendance and possibly a better future for individuals and communities.

Water quality can be considered as a measure of the suitability of water for its intended use, in terms of physical, chemical or biological characteristics (1). Water quality is very important since poor water quality can have adverse effects on human life. As stated earlier, any substance that alters the properties of water so it is no longer beneficial, safe or threatens the function of the ecosystem is considered a pollutant or contaminant. Contaminants in water may have the following effects on people:
Other than direct health effects, poor water quality can impact economic and social conditions around the world that can affect the quality of life. As emphasized by World Bank Group president David Malpass, “Clean water is a key factor for economic growth. Deteriorating water quality is stalling economic growth, worsening health conditions, reducing food production, and exacerbating poverty in many countries,”. Economic growth is intertwined with the quality of the environment, especially water, so much so that the World Bank estimates that water pollution can reduce economic growth by up to a third (3).
Suffice to say that water quality is of the utmost importance. This makes water monitoring and testing a significant step in assessing water quality. Around the world, there is a focus on the presence and monitoring of microbiological contaminants in the form of microorganisms in water because the health effects of these are the most pronounced.

Microorganisms or microbes in general are small living things with dimensions in the micron (micrometers) range or smaller. Microbes are roughly ten times smaller than the average thickness of the human hair and cannot be seen without the help of microscopes. Not all microorganisms are harmful; in fact many microorganisms around us help us and the environment to stay healthy. The microorganisms that are capable of causing harm to their hosts are called pathogens. This harm can be caused in the form of competing with the host for metabolic resources (food), destroying the host cells and tissues or producing toxins (1).
Pathogens can be categorized as:
Remember that not all microorganisms are pathogens. For example, yogurt is made by exposing milk to helpful bacteria (lactobacillus acidophilus).


Below are some of the harmful and dangerous pathogens that can exist in water and need to be monitored and tested in laboratories. The frequency of occurrence and the extent of danger requires some of them to be monitored and tested more regularly.
Escherichia coli (E.coli), Coliform* (fecal and non fecal), Legionella pneumophila, Salmonella, Campylobacter (C. jejuni & C. coli) (1) and Vibrio cholerae (V. Cholerae) (2)
*E.coli is a fecal coliform
Enteroviruses(such as poliovirus, echovirus, coxsackieviruses), Hepatitis A, adenovirus, astrovirus, E viruses, rotavirus, norovirus and other caliciviruses*.
*viruses that are excreted through the body via urine like polyomaviruses and cytomegalovirus can potentially be spread through water. Viruses like influenza and coronaviruses have been suggested to be transmitted through drinking water, but the current evidence is inconclusive (3).
Cryptosporidium, Giardia lamblia, Toxoplasma gondii, Entamoeba histolytica, Cyclospora cayetanensis, Isospora belli, Blastocystis hominis, Balantidium coli, Acanthamoeba spp., Sarcocystis spp. and Naegleria spp (4).
Fungi are not as heavily regulated in guidelines for water monitoring and are listed as “nuisance organisms” but this does not mean they are necessarily allowed in water. These are some of the species and genera of fungi in water (especially tap water) that can also be present in the air.
Aspergillus (e.g. mould), yeast of the gerena Candida, Exophiala, Fusarium, Malassezia, Ochroconis, Penicillium, Phialophora, Phoma and Rhinocladiella, Cryptococcus and Rhodotorula, and black yeast from the general Aureobasidium (5)
phytoplankton blooms, micro-algal blooms: Harmful Algal Blooms (HAB)
All these pathogens can cause mild to severe illnesses in humans if they exist in higher than Maximum Contaminant Levels(MCL) outlined in drinking water quality monitoring regulations*. Higher amounts of pathogens in waters that enter the environment can also hurt the ecosystem. In order to monitor the presence of these pathogens, indicators have been chosen to reflect a problem with water quality.
Indicators were chosen because they are:
The United States Environmental Protection Agency (USEPA) regulates turbidity, E.coli and Total Coliforms (fecal and non-fecal coliform) as these indicators (1). Bear in mind that turbidity is the cloudiness of water, not a microbe; but it can be a suitable environment for the growth of pathogens and disrupt disinfection. Total Coliforms can be found in the intestinal tract of animals, but not all coliforms are pathogens. They can come from fecal materials, soil, water or grains. Coliform bacteria belong to the Enterobacteriaceae family and include a number of bacterial species.
Although the Maximum Contaminant Level (MCL) for Total Coliforms is also non detectable per 100 ml of water, the revised rule proposed by the USEPA in 2010 eliminated MCL requirements for Total Coliforms and replaced it with MCL for E.coli as well as requiring monitoring, assessment and corrective action for the treatment systems in place for coliforms where there are treatment options available (7).
The following table includes the species, sources and illnesses caused by some of the most important pathogenic microorganisms in water.
| Pathogen | Source | Illness |
| Bacteria | ||
| E.coli | Sewage overflow, an Improperly working sewage system, Agricultural runoff, Stormwater runoff, Flooding (8,9) | Gastroenteritis, Pneumonia,Urinary tract infection |
| Legionella Pneumophila | Presence in water distribution systems, Construction, Water main breaks, Changes in municipal water quality, Biofilm, Scale and sediment, Water temperature fluctuations, pH fluctuations, Inadequate levels of disinfectant, Changes in water pressure, Water stagnation (10) | Legionellosis: Pontiac Fever, Legionnaires’ Disease |
| Salmonella | Sewage overflow, an Improperly working sewage system, Agricultural runoff, Storm water runoff, flooding (11) | Gastroenteritis, Salmonellosis, Typhoid, Reactive Arthritis |
| C. jejuni | Sewage overflow, Improperly working sewage system, Agricultural runoff, Storm water runoff, flooding12 | Gastroenteritis, Campylobacteriosis |
| Viruses | ||
| Poliovirus | Sewage overflow, an Improperly working sewage system (mainly from humans), Overland runoff, underground seepage (13) | Paralytic poliomyelitis |
| Rotavirus | Sewage overflow, an Improperly working sewage system (from humans), Stormwater runoff, Flooding (14) | Gastroenteritis |
| Hepatitis A. | Sewage overflow, Inadequately treated water and wastewater (15) | Liver inflammation |
| Protozoa | ||
| Cryptosporidium | Sewage overflow, an Improperly working sewage system, Agricultural runoff, Stormwater runoff, Flooding (16) | Cryptosporidiosis (may include fever, diarrhea and vomiting), More severe harm to digestive or respiratory tract in immunocompromised persons |
| Giradia Lamblia | Sewage overflow, an Improperly working sewage system, Agricultural runoff, Stormwater runoff, Flooding (17) | Giardiasis |
| Fungi | ||
| Aspergillus | Inadequately treated water or wastewater and the presence of organic materials, pH change, Water temperature fluctuations, Changes in water hardness, Changes in chemical composition of water (6,18) | Aspergillosis |
| Fungi from the genus Exophiala | Sewage overflow, Inadequately treated water or wastewater and the presence of organic materials, pH change, Water temperature fluctuations, Changes in water hardness, Changes in chemical composition of water (6,18) | Traumatic cutaneous infections, Keratitis, Onychomycosis, Otitis externa, Lung infections in patients with cystic fibrosis, Disseminated mycosis in immunocompromised patients, even involving the brain (6) |
| Algae | ||
| Phytoplanktons causing HAB | Nutrient pollution in the form of excess nitrogen and phosphorus caused by: Agriculture, Stormwater, Inadequately treated Wastewater, Fossil fuels, Household runoff (19) | Eye, skin, nose, throat and respiratory irritation (20), HAB toxin poisoning from eating contaminated seafood (21) |

To reiterate, there are different types of bacteria that can exist in water.
Escherichia coli (E.coli), Coliform (fecal and non fecal), Legionella pneumophila, Salmonella and Campylobacter (C. jejuni &C. coli) are some of the species that can find their way into water and cause different illnesses in animals and humans.
As mentioned, the most common ways for these bacteria to end up in water are inadequate water treatment, sewage overflow, improperly working sewage system, agricultural runoff, storm water runoff, flooding and in some cases changes in the physical and chemical properties of water.
Specific to drinking water, faulty piping systems in the distribution network can lead to pathogens in water. Pathogenic bacteria can be responsible for a variety of diseases like gastroenteritis or infectious diarrhea, pneumonia, urinary tract infection, legionellosis, typhoid, campylobacteriosis and cholera that may unfortunately lead to loss of life. As previously stated, dirty water causes the death of one human being every 10 seconds.
Below, we’ll focus on some of the bacterial species in water and their adverse effects, followed by information on water management. Water management is paramount in preventing waterborne diseases. Water treatment as well as water quality monitoring including water sample testing are the most effective forms of water management.



Escherichia coli (E.coli) are a large group of Gram negative*, rod shaped bacteria with about 1-2 micrometers in length and 0.5 micrometers in radius. E.coli bacteria are naturally found in the environment and the intestines of humans and animals. They can also be found in food and water.
Some strains can cause urinary tract infection and pneumonia. Some pathogenic E.coli belong to the group of bacteria called STEC (Shiga toxin-producing E.coli) also known as VTEC (Verocytotoxin-producing E.coli) or EHEC enterohemorrhagic E.coli. The most commonly identified strain of STEC is E.coli O157:H7; O157 and H7 refer to the antigens present in this strain1,2,3,4. E.coli O157:H7 is a hazardous strain although there are several other strains of pathogenic EHEC as well as other pathotypes of E.coli(not belonging to EHEC) that cause sickness.
Transmission usually occurs through coming in contact with the pathogen through ingestion via food, drinking contaminated water, contact with cattle, contact with the feces of infected people, and the like.
Since E.coli exists in the digestive tract of humans and animals, it can end up in different bodies of water from feces, including sources of drinking water such as rivers and lakes or private wells through sewage overflow, improperly working sewage system, agricultural runoff, storm water runoff and flooding .
Even small amounts of E.coli in water can be dangerous and lead to illness. Therefore the allowable amount or MCL in drinking water is non-detectable/absent in 100 ml.
Some international guidelines specify the MCL as zero in 100 ml of drinking water (5).
Extensive research has been done on the survival of E.coli in water and the conclusion is that since many factors such as temperature, sunlight and nutrients can affect the survival of E.coli, estimates and models are made for various conditions. Further field studies will help future estimates and calculations. For example, at a temperature of 14-20°C E.coli survives for less than 1-10 weeks in natural surface waters. In groundwater it can survive for 3-14 weeks at 10°C (6,7).
The dependence of the survival of E.coli on various environmental conditions means that regular and frequent monitoring, testing and assessment of water quality is crucial when it comes to E.coli to ensure the safety of water for humans and the environment.
*Gram negative bacteria have a particular substance called peptidoglycan in their cell wall that lose the crystal violet stain in the Gram staining method.
https://www.unwater.org/water-facts/water-sanitation-and-hygiene/.

Coliform bacteria belong to the family of Enterobacteriaceae. They are also Gram-negative, rod shaped bacteria like E.Coli bacteria. According to the different Standard Method (SM) of testing for coliform, this group of bacteria may be defined as (stated by Health Canada (1)):
These definitions refer to three roughly equivalent groups of coliform including a variety of species of various genera. Total Coliforms consists of non-fecal and fecal coliforms, which means whether or not the bacteria originate from feces.
Non-fecal coliforms normally occur in nature and are found everywhere. Not all coliforms are pathogenic. The following table includes some genera of coliforms (1,2).
| Coliform bacteria | Fecal | Non-fecal |
| Escherichia |  |  |
| Enterobacter |  |  |
| Klebsiella |  |  |
| Budvicia |  | |
| Leclercia |  | |
| Citrobacter |  |  |
| Pantoea |  | |
| Serratia |  | |
| Erwinia |  | |
| Hafnia |  |  |
The important role of Total Coliforms in water is that they are indicator organisms that can determine the presence of pathogens in water and its safety. As indicators Total Coliforms also help to assess the effectiveness of water treatment in different areas.
This is especially important for water sources used for providing drinking water.
Health Canada requires MCL or the maximum acceptable concentration (MAC) for Total Coliforms in water leaving a treatment plant and in non-disinfected groundwater leaving the well to be undetectable per 100 mL of water. This is also the case with WHO, the USEPA and EU (European Union) whenever water is disinfected.
Coliforms can survive in water from days to months depending on the environmental conditions. They are normally larger in numbers than other microorganisms in water. That’s one of the main reasons why they are perfect candidates for being indicators in water testing and quality monitoring.

source: en.m.wikipedia.org/wiki/Legionella#/media/File%3AHarmannella_entrapping_Legionella.png
According to WHO, species of the genus Legionella are Gram-negative, non spore forming, rod shaped, bacteria that are aerobic, which means they can survive and grow in an oxygenated environment. They have a non fermentative metabolism and require L-cysteine and iron salts to grow (1). Free-living legionellae are 0.3-0.9 micrometers wide and about 1.3 micrometers long. They can grow to 2-6 micrometers in vitro (outside the body) and are able to form filaments of 20 micrometers long.
Legionella bacteria are found naturally in freshwater environments. Small amounts of the bacteria in freshwater are not normally cause for concern and are not affiliated with illnesses; however, if Legionella finds its way into building water systems it can be a health risk. Some strains of Legionella like Legionella pneumophila can cause Legionellosis in the form of Legionnaires’ disease, which is a lung infection (pneumonia). The symptoms include coughing, fever, shortness of breath, muscle aches and Headaches. Legionellosis can also be a less severe illness in the form of Pontiac fever, which includes fever and muscle aches.
The below factors may lead to the growth of Legionella in building water systems (2,3):
For more information on where Legionella can grow or spread that may put humans at risk, visit the CDC webpage on Legionella.
There are no reports of person to person transmission of Legionella. Transmission commonly occurs by inhaling aerosols of contaminated water. In the United states, cases of Legionnaires’ disease have risen 350% from 2000 through 2016. The CDC states that it is unclear what the reason is for this increase but speculates it might be attributed to factors such as more awareness and more testing (representing an artifact in numbers increasing), increased susceptibility of the population, increased Legionella in the environment or a combination of such factors.
Although many countries do not have the means to report cases, WHO estimates that in Australia, Europe and the United States there are about 10-15 cases per million people (5). Regular testing, maintenance, cleaning and adequate disinfection of water and air conditioning systems, water cooling towers, manual and automatic faucets, water filters, etc will help to minimize the growth of the legionella bacterium as there are currently no vaccines available.
Bear in mind that not everyone is prone to getting infected. People, especially travelers over the age of 50, current or former smokers, people with chronic lung conditions, or are immunocompromised are at a higher risk (4).
Characteristically, legionellae multiply inside protozoa, but the presence of some cyanobacteria genera can promote their growth as well. In natural environments (except for thermal waters and water in tropical regions) legionellae concentrations fall below 1 cfu/ml* as their replication rates are low (1). In building water systems and water distribution systems where temperatures fall between 20°C and 50°C, legionellae frequently colonize. The bacterial cells can survive in aerosols for hours and last longer in water even in harsh conditions (6).
Due to their slow growth and metabolism, laboratory methods to test and detect Legionella are often slow processes that may take up to 14 days.
There are other methods such as biosensors or PCR (polymerase chain reaction) being developed to make rapid sensors for the detection of pathogenic Legionella such as Legionella pneumophila.
*cfu stands for colony forming unit, a measure of the concentration of microorganisms.


The word Campylobacter means “curved rod” from the Greek words kampylos (curved) and baktron (rod). This genus of bacteria belongs to the family Campylobacteraceae. Campylobacter bacteria are rod shaped, Gram negative, non spore forming, microaerophilic (require oxygen to survive but less oxygen than available in the atmosphere) and are non-fermenting. The cells are typically 0.2-0.9 micrometers in diameter by 0.5-5 micrometers in length and can grow to be as long as 8.0 micrometers.
The species C. jejuni is a common cause of food poisoning and it can be found in poultry and animal feces. Consumption of food or water contaminated with C. jejuni can cause Gastroenteritis and Campylobacteriosis which may induce symptoms like fever, diarrhea and abdominal pain. Campylobacter (C. jejuni and C. coli) is one of the leading causes of bacterial diarrheal disease in the world with 96 million cases in 2010 (1,2,3,4).
Together with E.coli, Legionella, Rotavirus and Norovirus, Campylobacter now makes up one of the leading causes of disease in humans(5) including contaminated water as one of the sources of disease.
Although a large portion of the cases of Campylobacteriosis are caused by contaminated food, water can also be contaminated with Campylobacter like C. jejuniand C. coli through the feces of animals. Sewage overflow, improperly working sewage system, agricultural runoff, storm water runoff or flooding can introduce feces into water. Transmission of disease other than through contaminated water is caused by consumption of contaminated food or coming in contact with contaminated feces from animals, especially poultry, or people.
Many factors such as temperature, concentration of dissolved organic matters and dissolved minerals can affect the survival of Campylobacter in water.Therefore the survival of the bacteria depends on the conditions of the body of water with studies showing some strains surviving 80 day in artificial water (6).


Salmonella are rod shaped, Gram negative, non spore forming, fermenting genera of bacteria from the family Enterobacteriaceae and are facultative anaerobes, which means they can grow in the presence of absence of oxygen. Salmonella bacterial cells are usually 0.7-1.5 micrometers in diameter and 2-5 micrometers long (1). Species of Salmonella like S. enterica (with almost all pathogenic Salmonella belonging to the subspecies enterica) are responsible for Gastroenteritis, Salmonellosis, Typhoid and even reactive arthritis after the infection has ended.
Salmonella is one of the most common causes of food-borne illness mainly in developed countries; it is also a major cause of diarrheal diseases in developing countries (1). In fact, Salmonella is 1 of the 4 key global causes of diarrhoeal diseases (2). Sewage overflow, an improperly working sewage system, agricultural runoff, storm water runoff and flooding can be the cause of Salmonella presence in water.
Some serotypes (type based on microbial surface) are only found in specific animals or in a single place. Others are found in a variety of animals and all over the world (4).
Transmission of disease can be through contaminated food and water or coming in oral contact with contaminated feces from humans or animals. Salmonella can survive for months in water depending on environmental conditions (2).

Now that we know the dangers of pathogens in water, we should discuss water management and measures to ensure the safety of water.
Water management, part of the broader concept of water cycle management, is planning and managing water resources for optimal use. It’s goal is to protect the lives of humans, animals and the environment. One of the aspects of water management is water quality management. It includes planning, developing and managing:
to minimize harm to humans, animals and the environment. These steps will ensure that water is safe for use and the quality of the wastewater that is distributed back into the water source like rivers and lakes has an acceptable level of pollutants.
Next we focus on the topics of water treatment and water quality monitoring.

Water treatment is the process of improving the quality of water for its intended use. It includes completely removing or reducing the concentration of various pollutants. The two main types of water treatment are drinking water treatment and wastewater treatment.
Drinking water treatment prepares safe drinking water for human consumption.
Wastewater treatment prepares water so that it is safe to be released into the environment and in more advanced treatment processes recycled for other purposes.
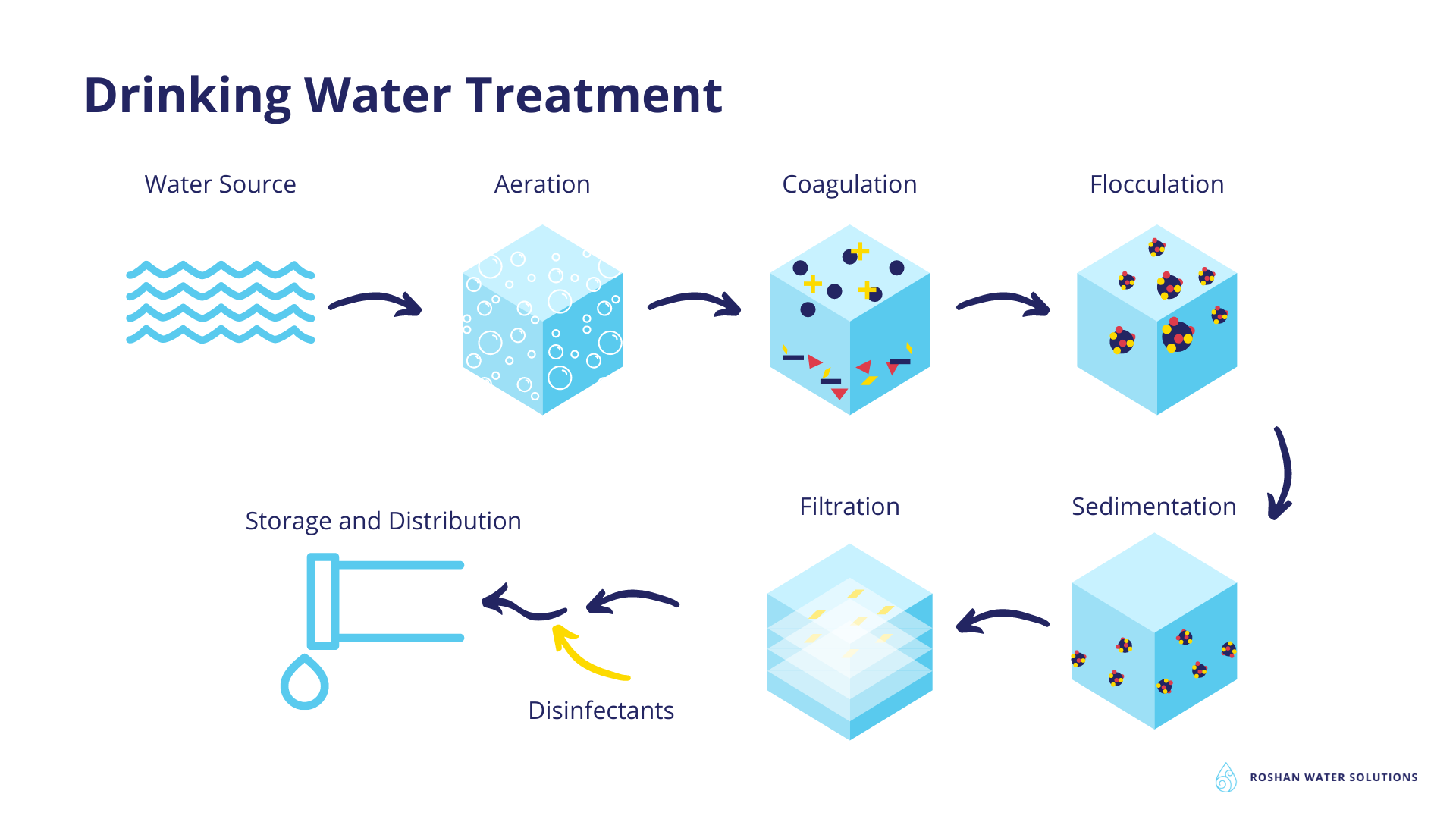
Public drinking water systems use different steps to remove the various bacterial contaminants (mentioned above) and provide safe drinking water to consumers. These steps commonly include:
In the coagulation and flocculation step, chemicals with positive electrical charge are added to water. Dirt and dissolved particles are negatively charged and repel each other. The added chemicals will help to overcome those repelling charges and neutralize the negative charge. This way the dirt and dissolved particles bind to the chemicals and form bigger particles called floc. Before the coagulation step, aeration is often used to remove dissolved gases and some metals through oxidation. In the aeration process air is introduced to thin layers of water or bubbles are introduced into the water.
Sedimentation involves the settlement of floc at the bottom of the water in the sedimentation tank because they are heavy. 27 to 84 percent of viruses and 32 to 87 percent of bacteria can be removed through coagulation, flocculation and consequent sedimentation because they are attached to the dissolved particles.
During the filtration process, the top water in the sedimentation tank that has become more clear will move through a series of filters made from sand, gravel and charcoal with different pore sizes. These filters will remove smaller particles like dust, some microorganisms and chemicals.
The previous steps in drinking water treatment cannot completely rid water of any remaining biological contaminants like bacteria, viruses and protozoa. That is why the last step in the drinking water treatment process is disinfection using disinfectants such as chlorine or chloramine; this disinfection is often called chlorination. The disinfectants are added to filtered water and the disinfected water leaves the treatment plant for distribution (1,2).
More recently, Ultra Violet (UV)-based treatment units have replaced the chlorination process as a disinfection step. This is mostly done due to the potential concerns over the formation of disinfection by-products during the chlorination of water that can potentially have adverse health effects like causing cancer and negative environmental impact (3). However, installation of UV treatment units increases both the capital expenditure and operational expenditure of a water treatment plant. This is why chlorination is still the disinfection method of choice for many water treatment plants.
In general, the quality of the water that enters the treatment plant determines the extent of water treatment. Surface water needs more treatment and filtration than groundwater since surface water in lakes, rivers, and streams contains more sediment and pollutants than groundwater. Surface water is also more likely to contain contaminants than groundwater (1,2).
The environment has an incredible ability to deal with small amounts of pollution and waste in water, but it cannot cope when dealing with an overwhelming amount of waste (4). 80% of the world’s wastewater, mostly untreated, is released into the environment (5). Moreover, 159 million people drink water directly from untreated surface water sources like streams or lakes (6). In addition, humans use surface water for recreational use. Animals use water to survive. Rivers, oceans and lakes are filled with aquatic species that rely on clean water. That is why treatment of wastewater is crucial for humans and the environment. Since the source of wastewater (e.g. sewage), its pollutants and its purpose differ from drinking water, the process of wastewater treatment is also different.
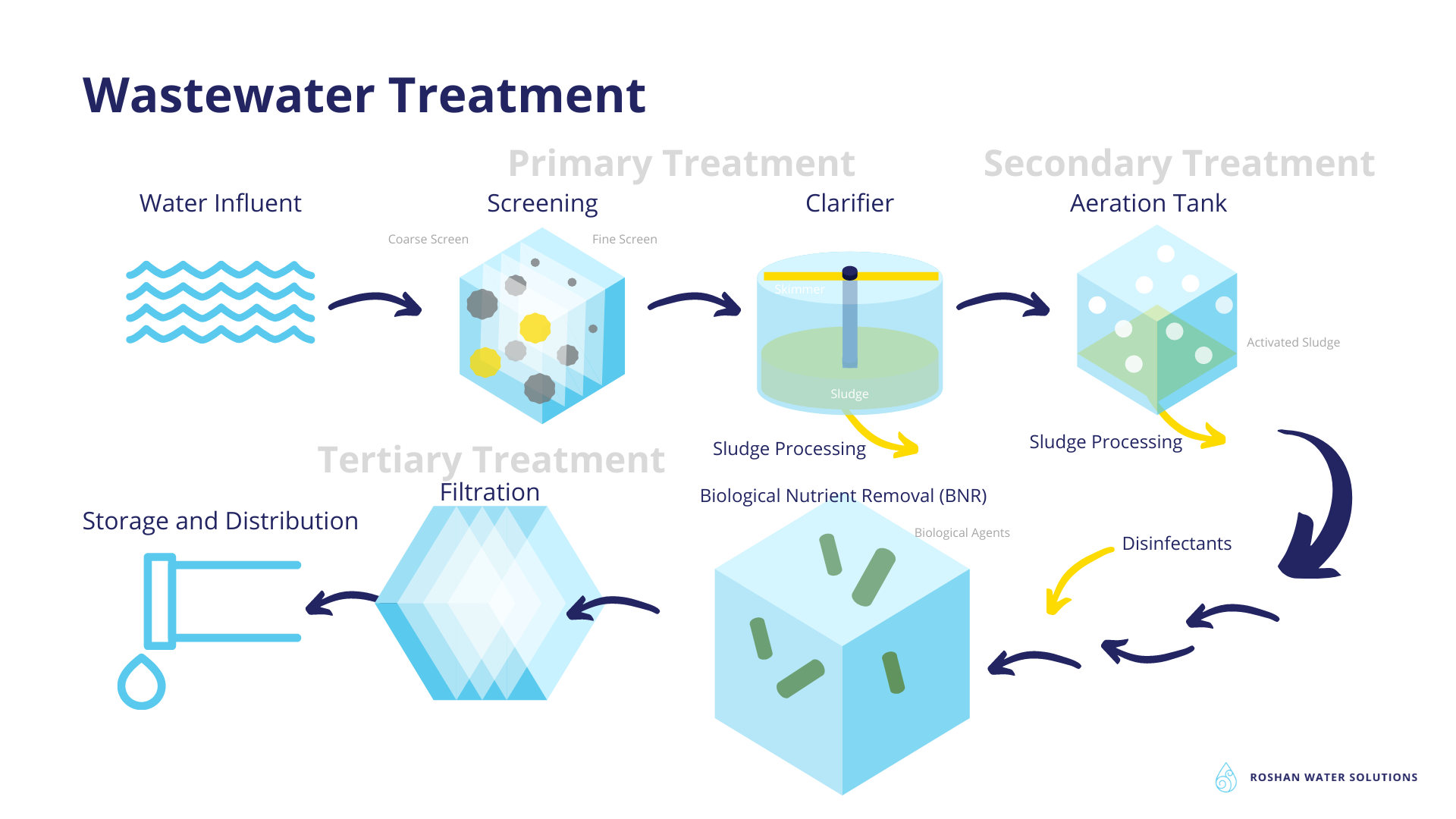
The type of wastewater treatment process depends on the volume of the wastewater. In general wastewater treatment may consist of primary, secondary and tertiary treatments. Bear in mind that some of these processes are essential regardless of the treatment plant size. Most municipal wastewater treatment facilities implement primary and secondary levels of treatment. Many treat wastewater further via tertiary treatments. The type and the order in which wastewater is treated varies in different treatment plants (7,8).
The primary treatment step consists of physical or mechanical processes for wastewater treatment such as sedimentation, screens, sieves or thermal methods.
Solids are firstly removed from wastewater. These solids contain up to 35% of the pollutants that need to be removed. Large solids like garbage, sticks, leaves or paper are omitted using screens that have openings of about 1cm.
After larger items are removed, wastewater is kept in settling tanks, often called clarifiers, for several hours. This way scum will float at the top of the tank and sludge will settle at the bottom of the water. Sludge is eliminated from the bottom and scum is skimmed from the top. The sludge and scum that is removed can also be treated separately to make biofuels. Approximately half of fecal coliforms are removed in this step. The organisms that use the oxygen in the water are also removed therefore bringing the biological oxygen demand (BOD) down to 50 percent (8). The efficiency of a treatment plant is determined based on the extent to which it removes suspended solids, as well as BOD (9).
Secondary Treatment
Secondary treatment involves the use of bacteria with the help of oxygen to consume, break down and digest contaminants. Methods include aeration, anaerobic wastewater treatment, biochemical oxidation, sewage ponds (lagoons) or sludge digestion. The biological process is also referred to as the activated sludge process. This sludge refers to smaller sludge bits that weren’t scraped in primary treatment. Secondary treatment is able to eliminate 85 to 90 percent of BOD and suspended solids. Most of the coliform bacteria, 90 to 95 percent, are removed as well. In some wastewater treatment facilities a sand filter is used to remove additional pollutants. Then the water goes through disinfection with chlorine, ozone, or UV before discharge.
Tertiary Treatment
Tertiary treatment is often referred to as advanced wastewater treatment since
not all treatment plants have the capability of using them. In tertiary treatment, physical, chemical and biological processes are utilized to remove additional pollutants like organic matter, metals, dyes and nutrients, namely phosphorus and nitrogen. Some tertiary treatment processes include biological nutrient removal (BNR), nitrification-denitrification, advanced filtration methods ranging from ultrafiltration, micro-filtration, nano-filtration and reverse osmosis. Secondary clarifiers can also be used as tertiary treatment.
The BNR process uses bacteria to digest pollutants in different tanks with different oxygen levels. Phosphorus is removed in this manner and ammonia breaks down to nitrate and nitrogen gas.
Smaller communities sometimes use lagoons or even septic tanks for primary treatment before they release wastewater into the environment. The water is given time to settle. Where land is available and water cannot flow to nearby bodies of water, biological agents in the soil can help to break down the contaminants in the wastewater. At times these methods may be inadequate for wastewater treatment. Shallow lagoons in particular do not allow enough space for settlement of all pollutants and are not suitable for long term storage of wastewater (8).

According to the United States Environmental Protection Agency (US EPA), water quality monitoring can be defined broadly as the sampling and analysis of water for its various constituents and conditions (1). These constituents may include:
As mentioned previously, water quality monitoring is one of the main elements of a successful water management strategy. The concept of water quality monitoring can be used in different applications and for different purposes including:
There are a battery of tests to determine the quality of water. That is why “Standard Methods For Examination of Water and Wastewater” has been developed. This collection of more than 400 standard methods has been continuously used and updated since 1905 and is the fruit of the joint collaboration between three well-known technical societies:
The following table summarizes various parts of this collection and briefly describes the methods involved in each chapter.
| Part No. | Part Name | Description | Parameters tested |
| 1000 | Introduction | This chapter is intended to provide overall information regarding the methods described in the rest of the collection. General guideline about quality assurance strategies, data quality, collection and preservation of samples, laboratory occupational health and safety as well as waste minimization and waste disposal strategies have been provided in this chapter. However, in each of the following parts, the second chapter always describes the specific quality assurance/quality control measures. | Not Applicable |
| 2000 | Physical and Aggregate Properties | Methods described in this chapter are used to determine overall physical properties of water samples, regardless of what specific constituents are present in water. Physical properties are used in this chapter in contrast to chemical composition, radioactivity or microbiological properties. However, it is mentioned that in some cases, it is difficult to distinguish between physical properties and chemical properties as both are intertwined. For example, tests for determination of the taste of water samples are included in this chapter although taste is heaviliy dependent on the chemical composition of water. | Appearance Color Turbidity Odor Taste Flavor profile analysis Acidity Alkalinity Calcium carbonate saturation Hardness Oxidant demand/requirement Conductivity Salinity Floatables Solids Temperature Particle counting and size distribution Asbestos Oxidation-reduction potential Tests on sludges Abaerobic sludge digester gas analysis Dissolved gas supersaturation |
| 3000 | Metals | The effects of the presence of metals in water and wastewater covers the whole range from being essential to the growth of plants and animals, to damaging the treatment processes and finally, imposing toxicity to the users. Moreover, for some metals, the shift in the type of the effects can depend on the concentration. As a result, this part describes a variety of methods that can be used for detection and quantification of metals in water samples depending on the complexity of the water matrix as well as required level of sensitivity and precision. Methods described in this part can be divided into two general categories of colorimetric methods and instrumental methods. As the name suggests, colorimetric methods quantify the metals in a water sample by measuring the degree of color formation in the sample when one specific metal reacts with a specially designed reactant used in the testing process. For example, in colorimetric determination of aluminum in water, Eriochrome cyanine R is used as the reactant. Colorimetric methods are usually simpler and more cost effective compared to instrumental methods; however, they are prone to interferances of other water constituents with the reactant. For instance, phosphate is a known interfering agent in colorimetric detection of aluminum mentioned above. Instrumental methods include tests such as atomic absorption spectrometry, flame photometry, nductively coupled plasma emission spectrometry, anodic stripping voltammetry. The choice of method is heaviliy dependent on the specific metal being detected, complexity of the water matrix, as well as desired detection range. For example, flame atomic absorption methods are normally suited for detection range of 0.1-10 mg/l while more complicated methods such as inductively coupled plasma mass spectrometry can offer detection as low as 0.01 µg/l. | Aluminum Arsenic Calcium Chromium Copper Iron Lead Lithium Magnesium Manganese Potassium Selenium Sodium Strontium Vanadium Zinc Other metals (Only contain background information for other matals not included here and refer the reader to the appropriate methods) |
| 4000 | Inorganic Nonmetalic Constituents | As opposed to the previous part, this part of the standard methods deals with the detection and quantification of non-metalic inorganics. In general, this part describes classical wet chemical techniques and their more modern automated variants in quantification of nonmetalic inorganics (e.g. ion chromatography, continuous flow analysis, flow injection analysis, capillary ion electrophoresis) . Of particular importance are methods in quantifcation of constituents such as chlorine, nitrogen and phosphorusSince chlorination is one of the most used disinfection methods in treatment of drinking water, detection of chlorine and its residuals in the treated drinking water and the receiving distribution system is of utmost importance. The presence of residual chlorine (in pre-determined concentration range) in the distribution system can be used as a proxy for assuring the sanitary condition of the water. However, it does not and should not replace the use of microbiological tests described in part 9 (especially Total Coliforms and E.coli). | Boron |
| 5000 | Aggregate Organic Constituents | In general, determination methods of organic matters in water and wastewater can be categorized in two groups. This part of standard methods deals with methods that analyze samples for aggregate organics properties. This means that methods presented in this part are concerned with quantification of overall concentration of organic compounds that have common chemical characteristics, regardless of their individual composition. These methods have many applications in analysis of raw & treated wastewater samples since they can assess the efficiency of the treatment process. Methods for quantification of individual organic compounds are presented in the next part. Some of the most important methods described in this part are: -Total organic carbon and chemical oxygen demand for analyzing the total amount of organic compounds present. -Biochemical oxygen demand for assessing the fraction of organic matters that are biodegradable. -Oil & grease which are defined as all organic compounds that can be extracted from the sample by non polar solvents. | Biochemical Oxygen Demand (BOD) Chemical Oxygen Demand (COD) Total Organic Carbon (TOC) Dissolved organic halogen Auatic humic substances Oil & grease Phenols Surfactants Tannin and lignin Organic and volatile acids Trihalomethanes and other disinfection byproducts UV-absorbing organic constituents |
| 6000 | Individual Organic Compounds | Methods presented in this part are complicated methods for detection and quantification of individual organic compounds at very low concentrations. As a results, for each method, stringent QA/QC protocols have been prescribed. Pre-concentration methods have been instrumental in achieving low levels of detection of organic compounds in water samples. In general, before using methods described here, a pre-concentration step is required to extract organic molecules from a large volume of water sample. Then analysis is done on a much smaller volume of extracted sample. Close-loop Stripping Analysis (CLSA), purge and trap technique and Solid-Phase Micro Extraction (SPME) are the most well-know pre-concentration methods used for preparation of the extracts for analysis. The extracts can be analysed using Gas Chromatography (GC) or Liquid Chromatography (LC) connected to one of several Mass Spectroscopic (MS) detectors. | Volatile organic compounds including: -Methane -1,2-dibromoethane (EDB) and 1,2-dibromo-3-chloropropame (DBCP) -Trihalomethanes and chlorinated organic solvents -Disinfection byproducts: haloacetic acids and trichlorphenol -Disinfection byproducts: aldehydes Extractable base/neutrals and acids including: -Phenols -Polychlorinated biphenyls -Polynuclear aromatic hydrocarbons -Nitrosamines Carbamate pesticides Organochlorine pesticides Acidic herbicide coumpounds Glyphosphate herbicide Tributyl tin Pharmaceuticals and personal care products |
| 7000 | Radioactivity | Radioactivity in water and wastewater samples can stem from natural occurrence as well as human activities. Human activities that can cause the contamination of water and wastewater with radioactive compounds can range from medical and industrial use of radioisotopes to mining and processing of nuclear fuel-related material and atmospheric testing of the nuclear devices. Testing protocols of water and wastewater samples for radionuclides can range from simple gross alpha and gross beta sreening to more sophistacted methods such as gamma spectroscopy. Gross screening measurements can be inexpensive, simple and quick. This is why these screening methods are normally used to obtain initial assessment and to determine whether further testing of the samples is required. However, these screening methods are subject to bias especially if a high concentration of dissolved solids is present in the sample. Moreover, they do not provide the isotopic composition of the sample and cannot be used to estimate radiation does. To determine the isotpoic composition and radiation dose, more complicated procedures such as gamma spectroscopy is required after the initial screening assessment. | Radioactive cesuim Radioactive Iodine Radium Radon Total radioactive strontium and strontium-90 Tritium Uranium |
| 8000 | Toxicity | Toxicity tests are important parts of water and wastewater monitoring programs since chemical and physical tests described in previous parts alone do not provide information on the potential effects of contaminations on aquatic biota. Moreover,when exposed to the same level of toxic compounds, the level of susceptibility vary between various aquatic species. Toxicity tests may be used to determine: – Suitability of environmental conditions for aquatic life – Effect of environmental parameters on the level of toxicity of compounds – Toxicity of specific wastes to specific test species – Relative susceptibility of aquatic species receiving a potentially toxic effluent – The effluent discharge rate allowed to be received by an aquatic environment | Mutagenesis Bacterial bioluminescence P450 reporter gene response to dioxin like organic compounds Comet/single-cell gel electrophoresis assay for detection of DNA damage Sediment porewater testing Algae Biostimulation (algal productivity) Phytoplankton Marine macroalgae Aquatic flowering plants Duckweed Aquatic emergent plants Ciliated protozoa Rotifers Annelids Mollusks Arthropods Daphnia Ceriodaphnia Mysids Decapods Aquatic insects Echinoderm fertilization and development Fish Fathead minnow Amphibians |
| 9000 | Microbiological Examination | The methods described in this part of the Standard Methods provide information about the microbiological content of water samples. Since microbiological content of samples are sensitive to the sampling procedures and the sample transportation conditions, the first five chapters of this section are devoted to precise QA/QC procedures, exact laboratory equipment needed for the test methods, how to prepare the equipment through adequate washing and sterilization, preparation of the culture media for methods using such media and, proper sampling and handling. Of particular importance are methods for testing water samples for Total Coliforms and E.coli. This is due to the fact that both Total Coliforms and E.coli are principal indicators of the suitability of water for domestic, industrial or other uses. Test methods (SM 9221, SM 9222 and SM 9223) for detection and quantification of these indicators in water and wastewater samples have become routine methods in any environmental laboratories that test water and wastewater samples, especially drinking water samples. These methods will be explored in more details in the next section of this article. This part of the Standard Methods also includes test procedures for differentiating various species in the Total Coliforms group. However, for drinking water applications, the differentiation of coliforms is of a very limited use since the presence of the Total Coliforms in a drinking water sample renders that water source unsuitable for use, regardless of the exact species present. Although it has to be mentioned that differentiation methods for drinking water can yield valuable information about the source of the pollution or how the distribution system is colonized. Heterotrophic Plate Count (HPC) methods are also included in this part. These methods provide an approximation of viable bacteria concentration in a water sample without identifying the types. These methods can be used in water treatment plants as in-house testing of the efficiency for various treatment processes or in standard laboratories for testing the quality of the reagent-grade water system. Test methods for isolating certain pathogenic bacteria or protozoa are also described in this part. These methods are normally very complicated and only used when detailed investigation of waterborne diseases or study of watershed are required. | Rapid detection methods Stressed microorganisms Recreational waters Heterotrophic plate count Direct total microbial count Assimilable organic carbon Aerobic endospores Multiple-tube fermentation technique for Members of the coliform group Membrane filter technique for members of the coliform group Enzyme substrate coliform test Detection of coliphages Differentiation of coliform bacteria Fecal enterococcus/streptococcus groups Iron and sulfur bacteria Nitrifying bacteria Detection of actinomycetes Detection of pathogenic bacteria Detection of enteric viruses Detection of fungi Pathogenic protozoa |
| 10000 | Biological Examination | The primary goal of this part of the standard methods is to describe protocols for field sample collection and laboratory analysis of water samples to establish the status of the aquatic species present and interpret the effect of current and past contaminations on these species. In other words, while previous parts of the Standard Methods deal with detection and quantification of various pollutants in the water, this part describes how those contaminants (such as turbidity or a chemical substance) can affect these aquatic communities. The aquatic species investigated here are extra to the ones mentioned in the previous section (see the parameter tested for this part). Some of the uses of the information produced by the test protocols in this part are: – To help interpret chemical analysis, for example, the presence or absence of certain biological species can point toward oxygent deficiency or supersaturation in water samples – To explain the source of taste, odor, color or visible particulates in the water – To help in design and operation of water and wastewater treatment plant by investigating the source of the clogging in pipes and filters – To evaluate the nature and extent of pollutions – To evaluate the status of self-putification in waterbodies | Plankton Periphyton Macrophytes Benthic macroinvertebrates Fishes Benthic meiofauna Nematological examination Identification of aquatic organisms |

As described in the previous sections, Standard Methods (SM) for Examination of Water and Wastewater provides a detailed explanation of various testing methods for determining the quality of water and wastewater by analyzing a variety of parameters, from metals to organic compounds and microorganisms.
Part 9 (SM 9xxx) is dedicated to all the methods for microbiological examination of water and wastewater samples. The table in the previous section lists all the microorganisms or parameters that can be tested with SM 9xxx. From all the methods in part 9, methods for detection and quantification of Total Coliforms and E.coli are of the utmost importance. As mentioned, this is due to the fact that Total Coliforms and E.coli are used as the microbial indicators in water quality management and represent the effectiveness of the utilized water or wastewater treatment systems in removing microorganisms.
As a result, millions of water (from both drinking water treatment units and the receiving distribution systems) and wastewater samples (usually treated effluents) are tested every year as parts of regular checks to make sure drinking water is safe for consumption and treated wastewater is suited for discharge into the environment.
In part 9 of the Standard Methods, three methods are dedicated to detection and quantification of Total Coliforms and E.coli. These are:
The following sections are only intended to provide a general overview of each standard method. More detailed information on specific protocols and procedures can be found in the Standard Methods reference document.
MTF is perhaps the oldest method of testing samples for Total Coliforms and E.coli. Fundamentally, the method works based on the fact that members of the Coliform group metabolize lactose. As a result, MTF utilizes lactose broth in combination with other compounds and detects the metabolism based on the formation of gas and acids in the test tubes. Gas formation can be observed by formation of visible gas bubbles in the tubes while the acid production changes the color of the medium.
MTF tests are done in three stages of presumptive tests, confirmatory tests and completed tests. It has to be mentioned that if in the presumptive stage, no gas formation or color change (due to acid production) is observed in any of the tubes, the sample is rendered safe, meaning confirmatory and completed stages are no longer required. If however, gas formation and color change are observed in the test tubes, water is unsafe and positive test tubes enter the confirmatory stage.
The following describes the procedure used in the presumptive stage.

Schematic showing the presumptive stage of MTF for untreated (polluted) samples
For treated samples:

For untreated samples:


As can be seen from this description, the MFT is a very complicated test method, bear in mind that the above-mentioned protocol is only for the presumptive stage. If the presumptive stage shows any positive test tubes, those tubes should enter the confirmatory and completed test stages which are even more complicated and out of the scope of the article. Suffice to say that during the completed test stage, a loopful of the samples in the positive tubes during the confirmatory stage are streaked onto plates containing highly selective medium such as Eosin Methylene Blue. A set of plates is incubated at 35 oC for Total Coliforms completed test results (formation of colonies with green sheen) while another set is incubated at 44.5 oC for thermotolerant E.coli completed results.
Beside being complicated, the MFT method is a lengthy test method that requires many glassware and media and can take up to 4-5 days to yield results.
The membrane filter technique was developed in the early 1950s in response to the fact that the MFT method described in the previous section is very labor intensive and time consuming. More importantly, as opposed to the MFT method that provides an estimation of the bacteria concentration using MPN tables, MF method gives the direct count of the bacterial colonies in the sample. This is simply why the MF method is one of the most used methods in the environmental laboratories for routine microbiological examination of water and wastewater samples.

Detection and quantification of Total Coliforms and E.coli by MF is done simply by the following steps.
There are multiple procedures described in SM 9222 for detection of Total Coliforms and E.coli. These procedures are more or less the same in the steps, with the main difference of compounds used in preparation of the selective medium are different. This change in the compounds dictates how the target bacteria is detected in the process. These procedures are:
Overall, the MF method is more versatile and simple compared to the MTF method and yields results that are more accurate. However, it cannot be used for samples with high turbidity.
Enzyme substrate methods for detection and quantification of Total Coliforms and E.coli in water and wastewater samples are the most recent addition to the Standard Methods.
The working principle of these methods is very similar to the Fluorogen/chromogen MI procedure used in the membrane filter method explained above where detection is done by the enzymatic action of bacteria on specific substrates in the medium. That action either produces color ro fluorescent signature. However, instead of counting colonies, the quantification principle is very similar to the MTF method where tests are done in a series of tubes or wells and statistical means are used to quantify the concentration of the target bacteria. As a result, similar to MTF methods, enzyme substrate methods provide the bacteria concentration with the Most Probable Number (MPN) unit.
In quantification of Total Coliforms, enzyme substrate methods use the chromogenic substrates known as ONPG and CPRG. In the test process, Total Coliforms produce the ß-galactosidase enzyme which hydrolyzes the substrates and produces a yellowish color. It is known that some other non-coliforms such as Pseudomonas can produce small amounts of the ß-galactosidase enzyme that can create false positives in the test. However, the medium includes compounds that suppress the activity of non-coliforms. As a result, false positives from these non-coliforms are generally avoided, unless they are present at very high concentrations (>106CFU/100 ml).
Quantitative analysis of E.coli is achieved by the fluorogenic substrates known as MUG. The ß-glucuronidase enzyme produced by E.coli cells reacts with this substrate and creates a bluish fluorescence under the UV irradiation. It is important to remember that the color change together with the presence of fluorescence in the sample indicates the presence of E.coli. This is because some strains of other species such as Salmonella can also produce fluorescence but since they lack the production of the ß-galactosidase enzyme, they cannot cause the color change.
IDEXX Colilert-18 Quanti-Trays are perhaps one of the most used technologies currently in the environmental laboratories that belong to the enzyme substrate method. In this procedure, 96-well trays are used that are similar to the image below.

100 ml of the water sample is added to the dry powder that contains all the necessary compounds for the reaction medium (chromogen, fluoregen, suppressants, etc.). The sample is shaken to dissolve all the chemicals and then is poured over the tray so all the 96 wells (48 large and 48 small wells). The tray is then sealed using a Quanti-Tray system and is kept in the oven at 35 oC for 24 hours. The following image shows the tray after incubation if Total Coliforms and E.coli are present.

The number of positive wells for each type of bacteria is then compared to standard tables and the concentration of Total Coliforms and E.coli is presented with the MPN unit.
Enzyme substrate methods are the simplest of the test methods for quantification of Total Coliforms and E.coli and they require lower expertise and equipment to be performed. However, they still require laboratory facility and cannot provide results in less than 24 h, similar to the MFT and MF methods discussed in this section.
The next section describes why new methods are still required to enable both onsite and rapid testing of water samples.

As described extensively in this article, water quality monitoring via regular testing of water samples is a cornerstone of any water management strategy and it is the first line of defense against any potential problems.
Regular testing of water samples for Total Coliforms and E.coli has become increasingly important since they show if water is safe for human consumption and whether the treatment units and the distribution system are functioning properly or not.
Standard tests used for detection and quantification of Total Coliforms and E.coli in water and wastewater samples were discussed in the previous section. Regardless of the complexity of each method, all three methods discussed in the previous section have two features in common. These are:
The fact that central laboratories are needed to perform the tests simply means that these tests are unavailable in many regions around the world, especially remote and small communities in developing countries. At the beginning of this article, we shared statistics about how these regions suffer from consequences of drinking unsafe water everyday.
The fact that current test methods need at least 24 hours to yield results means that even in regions around the world that have access to laboratory facilities, truly efficient water management strategies cannot be implemented since these strategies need real-time data as the input to enable real-time decision making.

Statistically, 25% of these boil water advisories are directly because Total Coliforms and E.coli were found in the water. Usually, boil water advisories are lifted after 48 hours on average, many of these boil water advisories can stay in effect for weeks, months and in some rare cases, years.
Alongside other deficiencies in the waterworks systems of some of these regions, boil water advisories are stretched because lifting them needs test data to make sure water is safe and tests are only done in central laboratories, which require time to provide the much needed data.
This is one of the many examples of why truly efficient water management strategies are in need of technologies that can provide real-time data so decisions can be made in real-time and onsite.
The above-mentioned discussion is precisely why we at Roshan Water™ Solutions have created VeloCens™. Although more information and specifications can be found on the VeloCens™ page, here we explain how our product solves the problems of 24 hour or more turnaround and central lab access requirements.
VeloCens™ is a portable device that quantitatively tests water samples for E.coli and Total Coliforms in 1 hour.
The fact that the device is portable means that tests can be done anywhere. Moreover, the test protocol is easy so anyone can do it. This means that the necessary data can be generated onsite and the nanotechnology used in the test cartridges, allows for test results to be ready within 1 hour. The following graph is included to help explain how VeloCens™ can do the tests rapidly and what the role of the nanostructures are in this process.

The bacterial growth curve shown in the above graph is a typical growth curve when bacteria (live) is incubated with enough nutrition to grow. The curve has 4 distinct phases:
All the current slow and laborious testing methods are heavily dependent on the exponential phase.
In this phase the population of the cells is so high that they form visible countable colonies on the membrane (for the MF method); or the metabolic activity is so significant (visible gas bubble formation in MFT) that it reacts with the substrates and imparts visible changes to the color or fluorescent properties of the solution (in enzyme substrate methods). For Total Coliforms and E.coli tests, the time to reach enough growth rate so the changes become visible is 24-48 hours. That is the reason why the current standard tests need this period of time to produce the results.
VeloCens™ also uses the same natural fundamental mechanisms in the cell growth to detect the target bacteria. However, instead of relying on the exponential phase, VeloCens™ works in the lag phase.In the lag phase, the cells still have their normal metabolic activity, however they are much smaller compared to the growth phase. The nanostructures used in VeloCens™cartridges are designed to be very sensitive to the metabolic activity of the cells.
As a result, even minute metabolism of the cells can be detected in the sample.
Finally, the engineering of these nanostructures, along with the blend of suppressants used in the test reagents impart the selectivity needed to deliver accurate test results in 1 hour.

Access to clean and safe drinking water is a global challenge. As a result it requires global initiatives and partnerships to solve it. The list of innovative companies such as Roshan Water™ who are trying to be part of the solution is endless. There are also massive local and global initiatives that are designed to solve water challenges in locations where clean drinking water is an everyday challenge.
Perhaps one of the most important initiatives in this category is currently being conducted by UNICEF called the Rapid E.coli Detection Project. The main goal of this program is for UNICEF to work with technology providers and “empower communities and government partners with key information on water quality”. This in turn will help these communities to treat unsafe water sources and identify areas where improvements are needed.
In this project, UNICEF is looking for technologies that allow testing water samples for E.coli to happen onsite and rapidly (within a few hours). One of the main applications for such technologies in this UNICEF project is performing household surveys of water quality in countries where UNICEF is providing support. UNICEF supports approximately 100 household surveys per year and on average each survey covers about 50,000 homes. This translates to roughly 1 million water tests per year to make sure the drinking water in these regions is safe for consumption.
There are many non-for profit organizations such as Humanity First and Ryan’s Well Foundation who actively conduct projects in rural areas in Africa, South East Asia, etc. with the goal to build water wells and hand pumps with enough sanitary conditions so the local population can have access to safe drinking water in their own community, without the need to travel for kilometers to reach one.

More than 2 billion people use a drinking water source that is contaminated with feces causing about 829,000 deaths per year due to unsafe drinking water. Diarrheal diseases claim the lives of roughly 5,000 children every day! The staggering approximately 1,200 deaths in children per day is linked to unsafe drinking water.
Overall, access to safe and clean water and assuring its safety is a global challenge. Monitoring the quality of water through testing is the first crucial step in resolving this challenge. Efficient strategies include decentralized, rapid testing for real-time decision making. VeloCens™ is a part of this solution for on-demand, onsite and rapid microbiological testing of water. It is a portable device that has the capability to quantitatively test water samples for E.coli and Total Coliforms in 1 hour.

Roshan Water™ has a long-term vision to deploy VeloCens™ in rural communities in developing countries to help people who are suffering the most from the consequences of drinking unsafe water.
